Overview
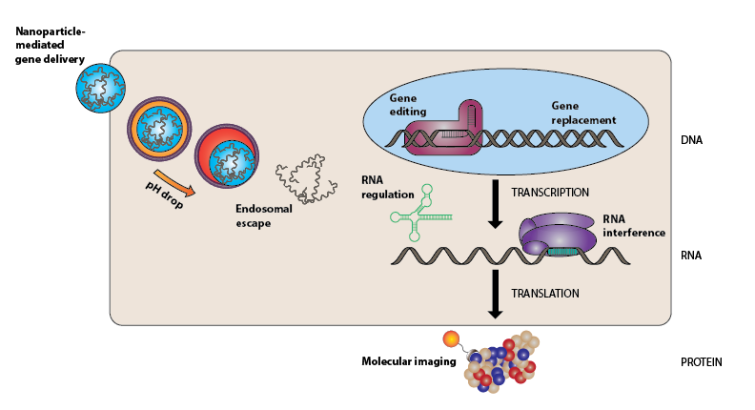
Case Center for Biomolecular Engineering seeks to develop cutting edge nanoplatforms for delivering nucleic acid therapeutics, and novel imaging agents to enable clinicians to better detect and alleviate the mortality and morbidity of life impairing or threatening diseases, including cancer. To accomplish this, we aim to mine signatures that are reflections of the tumor microenvironment, cellular or subcellular processes in the diseases. Further, our goal is to design and develop simple and smart biomolecules to target specific biological signatures for accurate detection and effective treatment of diseases.
Molecular Imaging
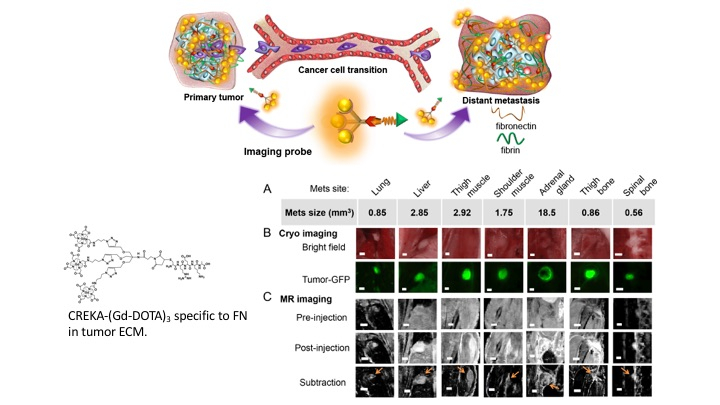
We focus on developing novel imaging probes capable of detecting and evaluating the aggressiveness of both breast and prostate cancer. Breast and prostate cancers represent the leading cause of morbidity and mortality in women and men, respectively. Using current diagnostic techniques, the diagnosis of these cancers suffers from high false-positive rates and relatively low detection sensitivity.
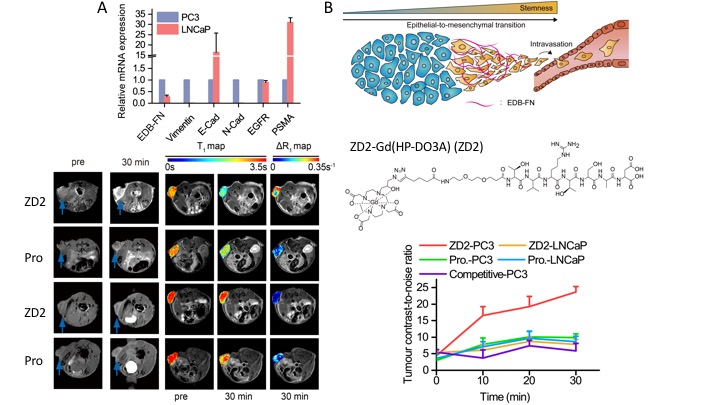
To overcome these hurdles, our research focuses on developing imaging probes specifically targeted to biomarkers in the tumor microenvironment, which is highly remodeled during cancer progression. We are particularly interested in targeting fibronectin, an extracellular matrix molecule that plays an important role in facilitating tumor progression. Fibronectin-targeted imaging probes developed in our lab demonstrated superior detection sensitivity and specificity in aggressive cancers with both MRI and PET when compared with clinical agents. These probes have great clinical potential to improve the detection, risk-stratification, preoperative imaging, active surveillance, and evaluation of therapeutic response of cancers.
Gene Therapy
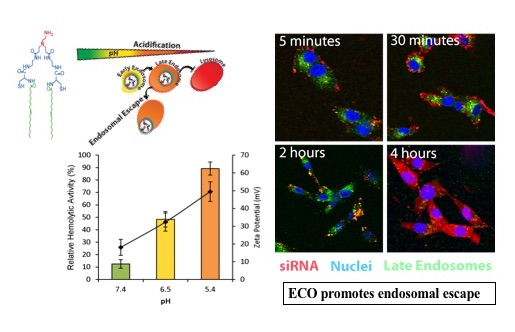
Our lab also focuses on the development of simple, safe, and smart multifunctional lipids that can efficiently deliver non-coding RNAs to cancer cells. One such example is ECO, a pH-sensitive multifunctional lipid carrier that has garnered attention for its clinical potential. It contains 1) a protonable ethylenediamine head group, 2) two cysteine based linker groups, and 3) two oleic acid lipid tails. Stabilization of the nanoparticles is achieved through the electrostatic condensation of the cationic head group with the negatively charged gene cargo, reversible disulfide formation through autoxidation of the cysteine groups, and hydrophobic condensation of the oleic acid lipid tails. Endocytic uptake of the nanoparticles is followed by pH-sensitive amphiphilic endosomal escape, and reductive cytosolic (PERC) release of genetic cargo.
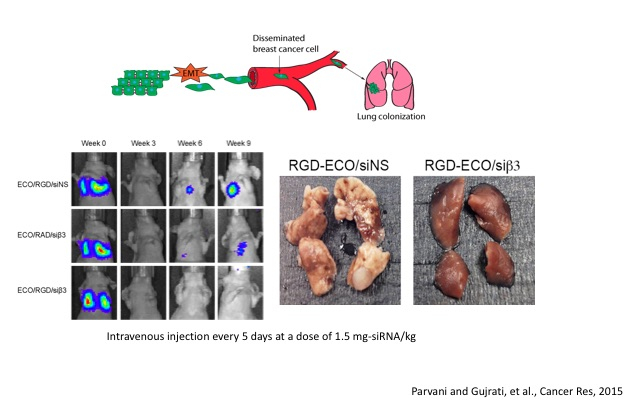
1) Silencing β3 integrin to inhibit Triple-negative breast cancer (TNBC) metastasis
We have utilized ECO to treat TNBC, a highly aggressive subset of breast cancer where patients exhibit an overall poor survival rate due to the development of metastasis and drug resistance. Metastasis has been associated with the biological programme known as epithelial to mesenchymal transition (EMT), where cells exhibit decreased cell adhesion properties and increased motility, allowing for successful invasion and dissemination to distant sites. β3 integrin has been shown to be essential for EMT and breast cancer metastasis, and we have shown that by utilizing ECO/siβ3 nanoparticles is effective in silencing β3 integrin, and attenuating EMT and invasion. Injections of ECO/siβ3 nanoparticles reduced tumor growth and inhibited metastasis in vivo. These results show ECO/siβ3 nanoparticles as a promising treatment for TNBC.
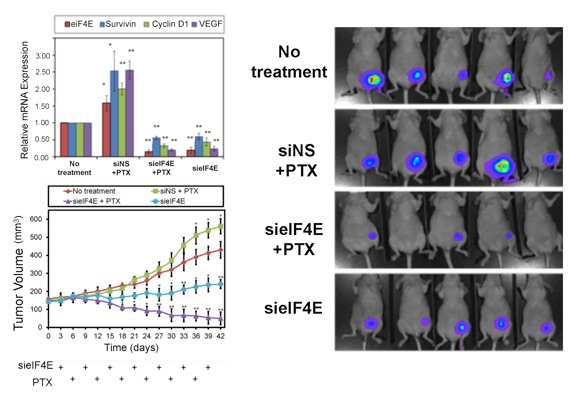
2) Silencing eIF4E alleviates paclitaxel resistance for TNBC therapy
Multidrug resistance (MDR), the mechanism where cancer cells become impervious to chemotherapeutic drugs, is a major challenge in cancer therapy. Knockdown of oncogenes specific to MDR has the potential to aide in the efficacy of current chemotherapy treatments. We have developed a dual pH sensitive lipid-siRNA nanoparticles, RGD-PEG(Hz)-ECO/siRNA, that effectively silences eukaryotic translation initiation factor 4E (eIF4E), an oncogene upregulated with paclitaxel resistance. Silencing of eIF4E re-sensitizes TNBC drug resistant cells to paclitaxel, both in vitro and in vivo, with negligible side effects. Significant tumor regression is also exhibited upon treatment with RGD-PEG(Hz)-ECO/sieIF4E every six days for six weeks.
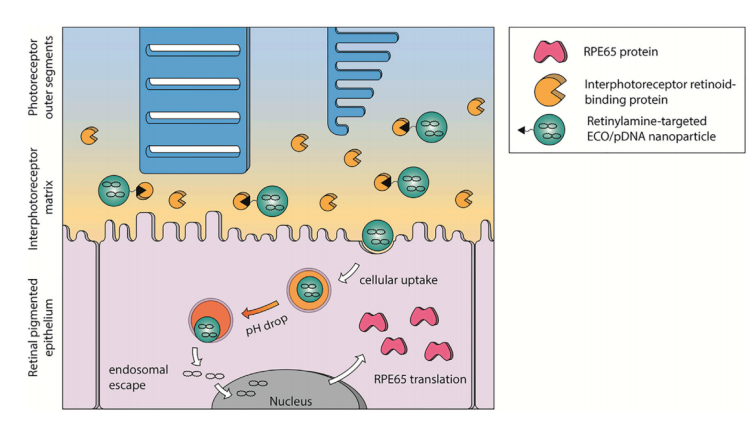
3) Multifunctional ECO Plasmid DNA nanoparticles for gene therapy of Leber’s Congenital Amaurosis (LCA) and Stargardt disease
Leber’s congenital amaurosis type 2 (LCA) is a rare genetic disease that causes severe and early vision loss due to retinal degeneration. One molecular form of this disease, known as LCA type 2 (LCA2), is caused by mutations in the RPE65 gene. These mutations result in an accumulation of al-trans-retinyl esters, thereby causing rod and cone photoreceptor dysfunction. Here, we utilized our lipid ECO-based delivery gene delivery system to transfect an RPE65 plasmid (pRPE65) into RPE65-/- mice. The nanoparticle was modified with all-trans-retinyl-amine to enhance the targeting to the RPE in vivo (Ret-PEG-ECO/pRPE65). We show that gene replacement therapy with Ret-PEG-ECO/pRPE65 nanoparticles produced an enhanced and sustained transfection efficiency in the RPE in vivo, and further improved the ERG activity of RPE65-/- mice for at least 120 days. The platform technology can also be used to deliver therapeutic genes to treat other monogenic retinal disorders, such as Stargardt disease.
4) New Research Projects
Our work currently focuses on exploring other non-coding RNAs for TNBC therapy, including microRNAs (miRNAs) and long non-coding RNAs (lncRs).