2026
We are proud to announce that two BEES2 faculty members have been elected to the National Academies. Congratulations to Edward Maginn for being elected to the National Academy of Engineering (NAE, 2026) and Jesse Wainright for being elected to the National Academy of Inventors (NAI, 2025).
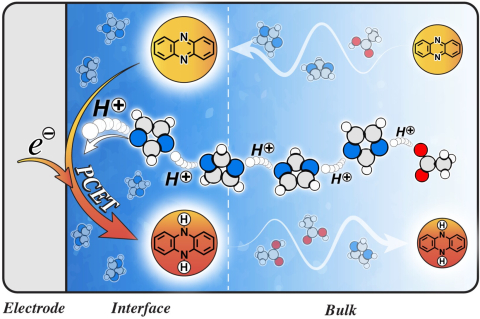
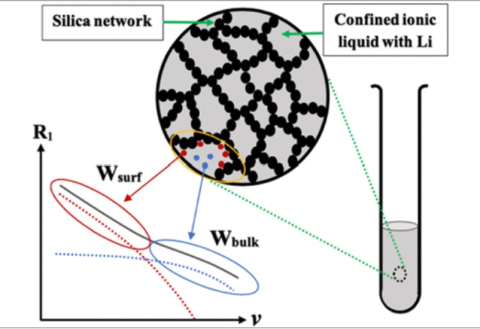
Miguel Muñoz, Michael S. Chen, Giselle de Araujo Lima e Souza, Thomas Simunovic, Vaishali Khokhar, Peisen Qian, Jesse Wainright, Robert Savinell, Andrew Parnell, Steven Parnell, Rachel C. Kilbride, Thomas A. Zawodzinski, Mark Dadmun, Steven G. Greenbaum, Joaquín Rodríguez-López, Mark Tuckerman, and Burcu Gurkan "Structured Electrolytes Facilitate Grotthuss-type Transport for Enhanced Proton Coupled Electron Transfer Reactions" PNAS, 2026
DOI: https://doi.org/10.1073/pnas.2530367122
2025
Case Western Reserve University hosted the annual meeting for the BEES2 Student Research Organization (SRO) on October 30th and 31st. The event brought together members for an engaging meeting focused on active collaborations.
Members of the BEES2 team attended the 2025 EFRC-Hub-CMS-CCS Principal Investigators meeting, sponsored by the Department of Energy’s Office of Basic Energy Sciences (DOE BES). The team presented their latest BEES2 research, and an illustration by Miguel Muñoz was honored with the “People’s Choice Award” at the Art of Science Image Contest.
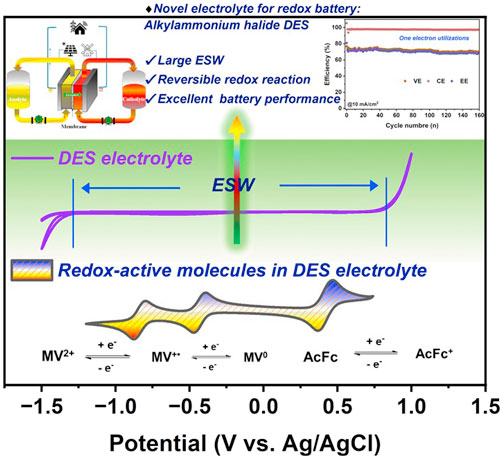
Zeru Syum, Jesse Wainright, Desiree Mae Prado, Burcu Gurkan, Palani Sabhapathy, Clemens Burda, Matthew Vasil, and Robert Savinell "Harnessing Redox-Active Molecules in Alkylammonium Halide-based Eutectic Solvents for Redox Flow Batteries" Journal of the Electrochemical Society, 2025
DOI: https://doi.org/10.1149/1945-7111/ade406
Brian A. Barth, Adam E. Imel, Thomas A. Zawodzinski " Electrochemical Processes Breaking Strict Phase Electroneutrality in Microemulsions" Journal of the Electrochemical Society, 2025
Vaishnavi Sree Jeganathan, Nicholas Sinclair, and Rohan Akolkar "Kinetics Measurements in Resistive Electrolytes Using Ring-Disk Electrode: Ring as Current “Shield” Enables Uniform Disk Current Distribution" ACS Electrochemistry, 2025
DOI: 10.1021/acselectrochem.4c00238

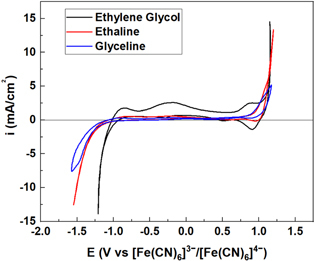
Rathiesh Pandian, Clemens Burda "Insights from Femtosecond Transient Absorption Spectroscopy into the Structure–Function Relationship of Glyceline Deep Eutectic Solvents" Molecules, 2025
DOI: 10.3390/molecules30051059
Desiree Mae Prado, Aaron N. Gonzaga, Brady Carter, Clemens Burda "Thermodynamic Water Activity Explains the Unusual Electrochemical Stability of Aqueous Deep Eutectic Solvents" Chemistry-A European Journal, 2025

Burcu Gurkan, Vaishali Khokhar, Sabhapathy Palani "Structured Electrolytes for Energy Storage" ACS in Focus, 2025
DOI: 10.1021/acsinfocus.7e9003
Case Western Reserve University was proud to host the entire BEES2 team for a productive all-hands meeting on March 10th-11th, 2025. The event brought together members to align on project goals and plan future milestones.
Palani Sabhapathy, Uddalak Sengupta, Miguel Munoz, Eda Cagli, Matthew T. Figgins, Jesse Wainright, David C. Powers, Burcu Gurkan "Conformational Control as a Design Strategy to Tune the Redox Behavior of Benzotriazole Negolytes for Nonaqueous Flow Batteries" ACS Applied Materials & Interfaces, 2025

Nicholas S Sinclair, Dinis Abranches, Robert F Savinell, Edward Maginn, Jess S Wainright, "Understanding Redox Organic Behavior in Deep Eutectic Solvents: Considerations for Molecular Design" Journal of The Electrochemical Society, 2025

Derrick Poe, Dinis O. Abranches, Xiaoyu Wang, Jeffrey Klein, William Dean, Benworth B. Hansen, Brian Doherty, Carla Fraenza, Burcu Gurkan, Joshua R. Sangoro, Mark Tuckerman, Steven G. Greenbaum, Edward J. Maginn, "Structural and Dynamic Heterogeneity of Deep Eutectic Solvents Composed of Choline Chloride and Ortho-Phenol Derivatives" The Journal of Physical Chemistry B, 2025

2024
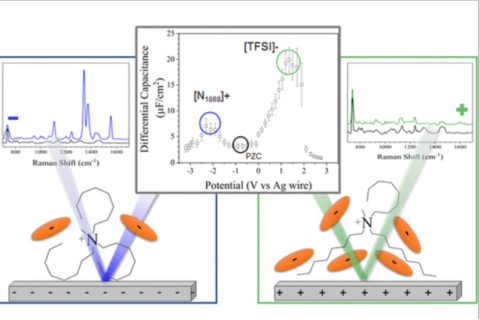
Shehan M. Parmar, William Dean, Changwoo Do, James F. Browning, Jeffrey M. Klein, Burcu E. Gurkan, Jesse G. McDaniel, "Structural Properties of [N1888][TFSI] Ionic Liquid: A Small Angle Neutron Scattering and Polarizable Molecular Dynamics Study" The Journal of Physical Chemistry B, 2024

Nicholas R. Furth ;Adam E. Imel; Thomas A. Zawodzinski "Comparison of Machine Learning Approaches for Prediction of the Equivalent Alkane Carbon Number for Microemulsions Based on Molecular Properties" The Journal of Physical Chemistry A 2024
Desiree M. Prado, Alan Robledo, Katherine Hightower, Anthony Jahng, Brian Doherty, Kayla Poling, Mark Tuckerman, Clemens Burda "Breakthrough Conductivity Enhancement in Deep Eutectic Solvents via Grotthuss-Type Proton Transport" Advanced Materials Interfaces, 2024
Giselle de Araujo Lima e Souza, Emilia Pelegano-Titmuss, Miguel Muñoz, Burcu Gurkan, Maria Enrica di Pietro, Andrea Mele, Phillip Stallworth, Steven Greenbaum, "Probing the potential of type V Deep eutectic solvents as sustainable electrolytes" Journal of Molecular Liquids, 2024
DOI: 10.1016/j.molliq.2024.126526

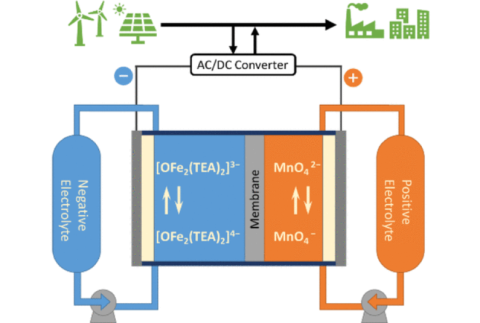
Xiaochen Shen, Christian Kellamis, Vincent Tam, Nicholas Sinclair, Jesse Wainright, Robert Savinell, "An All-soluble Fe/Mn Based Alkaline Redox Flow Battery System" Applied Materials and Interfaces, 2024
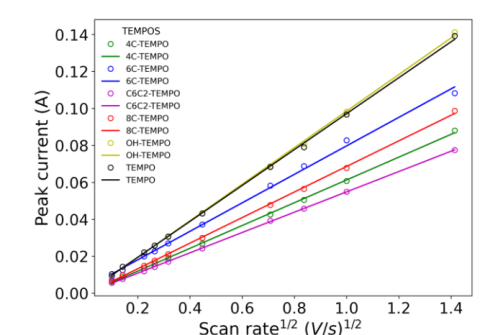
Maria Escamilla, Ernesto Camilo Zuleta Suarez, Hannah K. Davis, Jacob Johnson, Adam Imel, Brian Andrew Barth, Thomas Zawodzinski and Emily Pentzer, "Synthesis of Alkoxy-TEMPO Aminoxyl Radicals and Electrochemical Characterization in Acetonitrile for Energy Storage Applications" Journal of The Electrochemical Society, 2024
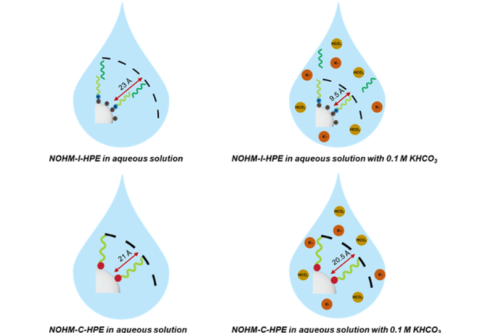
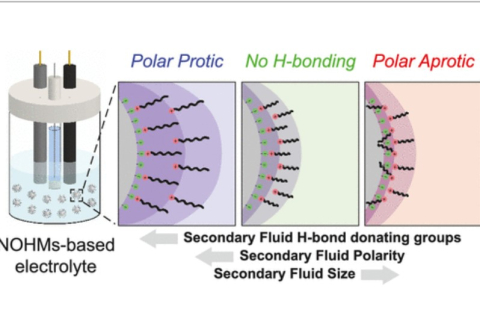
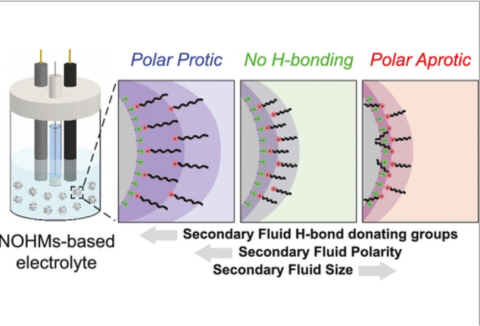
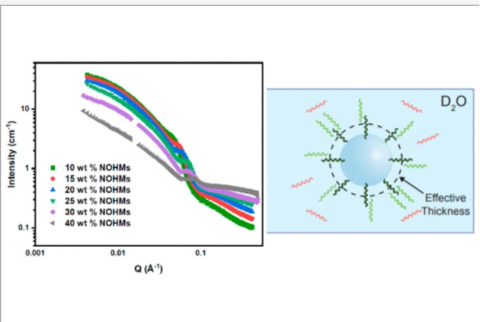
M. A. Haque, S.T. Hamilton, T. Feric, A.-H. A. Park, M. D. Dadmun, "Insights into the Assembly and Conformation of Nanoparticle Organic Hybrid Materials (NOHMs) in Solution with Varying Grafting Type" ACS Applied Polymer Materials, 2024
M. A. Haque, S.T. Hamilton, T. Feric, A.-H. A. Park, M. D. Dadmun, "Elucidating the assembly of Nanoparticle Organic Hybrid Materials (NOHMs) near an electrode interface with varying potential using Neutron Reflectivity" Nanoscale, 2024
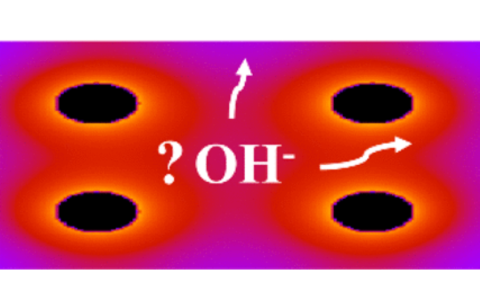
T. Zelovich, D. R. Dekel, M. E. Tuckerman "Electrostatic Potential of Functional Cations as a Predictor of Hydroxide Diffusion Pathways in Nanoconfined Environments of Anion Exchange Membranes" J. Chem Phys, 2024
Alfurayj, Prado, Prado, Samia, Burda, "Unusual Hydration Properties of Choline Fluoride-Based Deep Eutectic Solvents" J. Chem Phys, 2024
Gabriela B. Correa, Yong Zhang, Charlies R.A. Abreu, Frederico W. Tavares, Edward J. Maginn, "Revisiting the pseudo-supercritical path method: An improved formulation for the alchemical calculation of solid–liquid coexistence" J. Chem Phys, 2024
Coskun, Oguz Kagan; Munoz, Miguel; Dongare, Saudagar; Dean, William; Gurkan, Burcu, "Understanding the electrode-electrolyte interfaces of ionic liquids and deep eutectic solvents" Langmuir, 2024
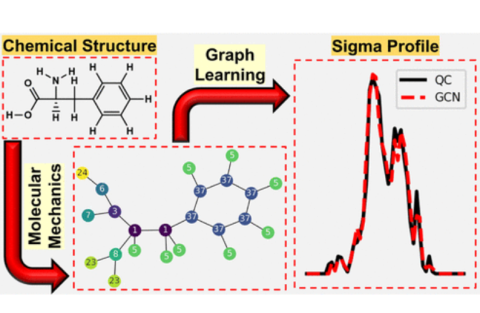
Dinis O. Abranches, Edward J. Maginn, Yamil J. Colón, "Boosting Graph Neural Networks with Molecular Mechanics: A Case Study of Sigma Profile Prediction" Journal of Chemical Theory and Computation, 2024
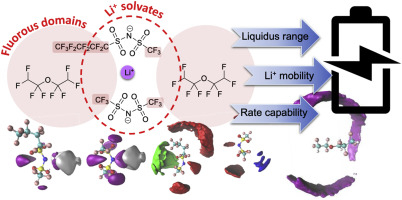
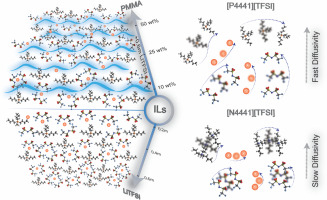
Drace Penley, Hazel Gerber, Mounesha Garaga, Nalinda Wickramasinghe, Steven Greenbaum, Edward Maginn, Yong Zhang, and Burcu Gurkan,"Impact of fluorination on Li+ solvation and dynamics in ionic liquid-hydrofluoroether locally concentrated electrolytes" Journal of Power Sources, 2024
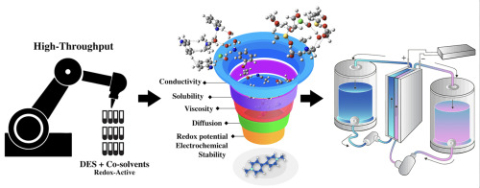
William Dean, Miguel Muñoz, Juran Noh, Yangang Liang, Wei Wang, and Burcu Gurkan "Tuning and high throughput experimental screening of eutectic electrolytes with co-solvents for redox flow batteries" Electrochemica Acta, 2024
Giselle de Araujo Lima e Souza, Maria Enrica Di Pietro, Franca Castiglione, Patricia Fazzio Martins Martinez, Maleen Middendorf, Monika Schönhoff, Carla Cecilia Fraenza, Phillip Stallworth, Steven Greenbaum, Alessandro Triolo, Giovanni Battista Appetecchi, and Andrea Mele "Unveiling the transport properties of protic ionic liquids: Lithium ion dynamics modulated by the anion fluorine reservoir" Electrochemica Acta, 2024
Giselle de Araujo Lima e Souza, Maria Enrica di Pietro, Franca Castiglione, Patricia Fazzio Martins Martinez, Carla Cecilia Fraenza, Phillip Stallworth, Steven Greenbaum, Alessandro Triolo, Giovanni Battista Appetecchi, and Andrea Mele "Polymer electrolytes based on protic ionic liquids with perfluorinated anions for safe lithium-ion batteries" Electrochemica Acta, 2024
Rathiesh Pandian, Daniel Kim, Yong Zhang, Ibrahim Alfurayj, Desiree Mae Prado, Edward Maginn, and Clemens Burda, "Chain length and OH-spacing effects on diol-based deep eutectic solvents" J. Mol Liquids, 2024
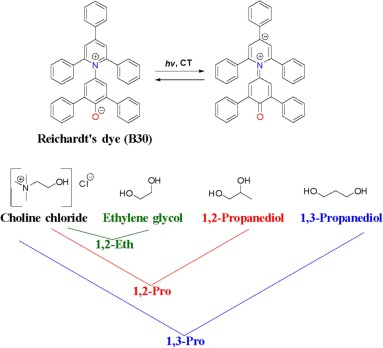
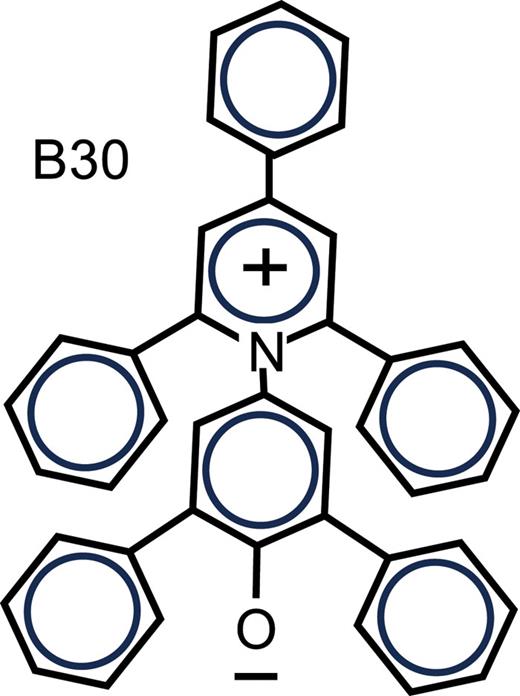
William Parson and Clemens Burda, "Calculated solvent reorganization entropies, free energies, and fluctuations of the energy gaps for intramolecular electron transfer and excitation of the solvatochromic dye B30" The Journal of Chemical Physics, 2024
Vaishnavi Sree Jeganathan, Nicholas Sinclair, and Rohan Akolkar, "Methods—Meso-Scale Electrodes for Characterizing Diffusion-Reaction Properties of Redox-Active Organics in Viscous Electrolytes" Journal of the Electrochemistry Society, 2024
2023
Stephanie Spittle, Ibrahim Alfurayj, Benworth Bryce Hansen, Kaylie Glynn, William Brackett, Rathiesh Pandian, Clemens Burda, and Joshua Sangoro, "Enhanced Dynamics and Charge Transport at the Eutectic Point: A New Paradigm for the Use of Deep Eutectic Solvent Systems" JACS Au, 2023
Ibrahim Alfurayj, Carla Cecilia Fraenza, Rathiesh Pandian, Steve Greenbaum, Clemens Burda, "Nuclear Magnetic Resonance Relaxation Pathways in Electrolytes for Energy Storage" International Journal of Molecular Sciences, 2023
Ibrahim Alfurayj, Carla Cecilia Fraenza, Rathiesh Pandian, Steve Greenbaum, Clemens Burda, "Solvation dynamics of choline fluoride in ethylene glycol – Water mixtures" Journal of Molecular Liquids, 2023
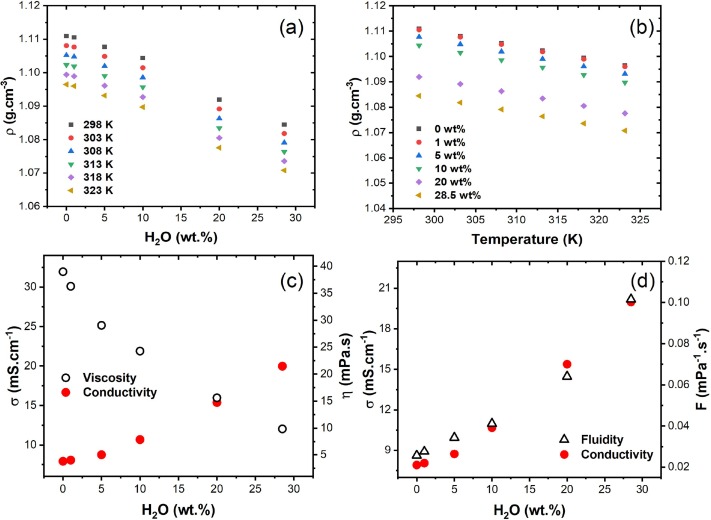
Kevin Echelberry, Henrik Burda, Preston Willis, William Parson, Clemens Burda, "Temperature-Dependent Solvent Reorganization Entropies, Free Energies, and Transition Dipole Strengths for the Photoexcitation of Reichardt's Dye B30" The Journal of Chemical Physics, 2023
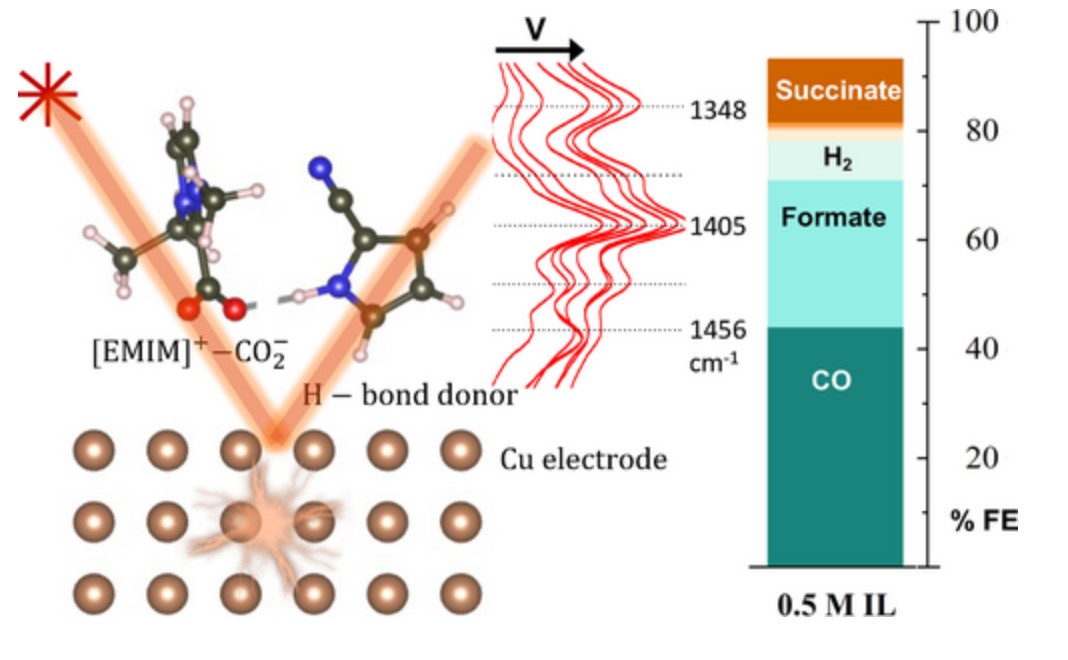
O. K. Coskun, S. Dongare, B. Doherty, A. Klemm, M. E. Tuckerman, and B. Gurkan, "Tailoring Electrochemical CO2 Reduction on Copper by Reactive Ionic Liquid and Native Hydrogen Bond Donors" ACS Central Science, 2023
Nora Shaheen, Bethany Kersten, Rohan Akolkar, "Mathematical Framework Underlying the In Situ Electrochemical Diagnosis of Adsorbed Intermediates Formed during Redox Reactions at Electrode Surfaces," Journal of the Electrochemical Society, 2023
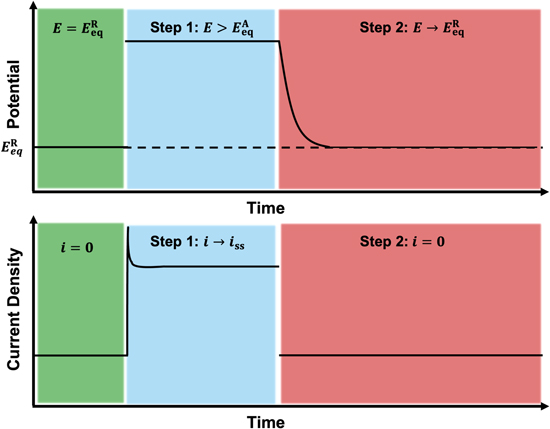
Desiree Prado, Xiaochen Shen, Robert Savinell, Clemens Burda, "Hydrodynamic Voltammetry of Fe2+/Fe3+ in Aqueous Deep Eutectic Sovents for Sustainable Redox Flow Batteries," Electrochemica Acta, 2023
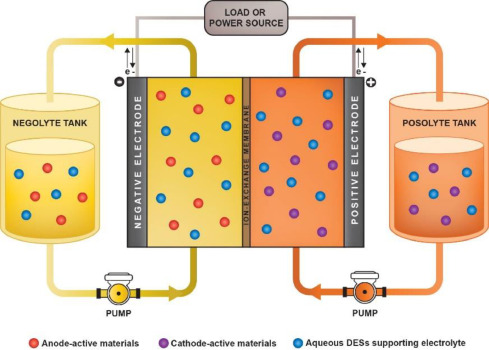
Ibrahim Alfurayj, Rathiesh Pandian, Sarah Springer, Clemens Burda, "Choline Flouride-Ethlyne Glycol Deep Eutectic Solvent Mixture-Synthesis and Physiochemical Properties," J. Mol Liquids, 2023

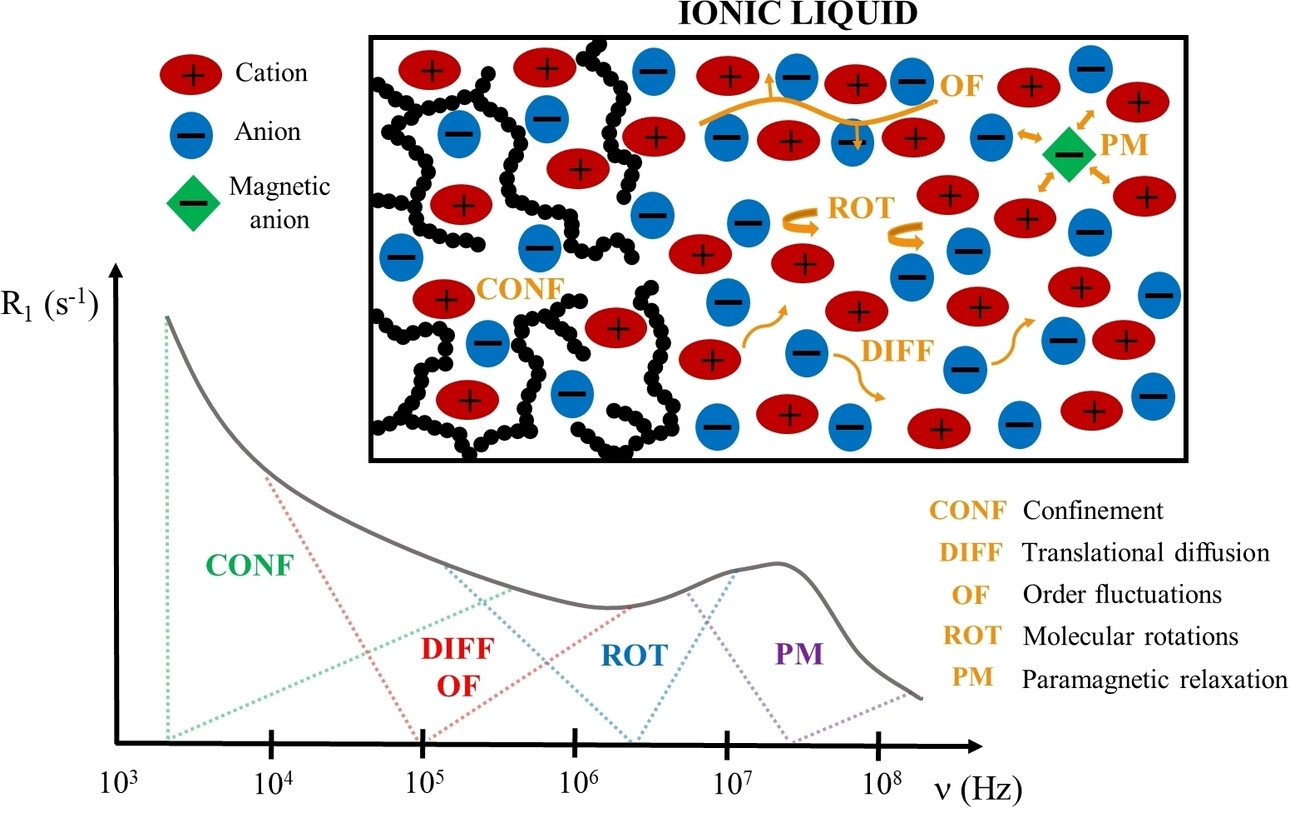
Carla C. Fraenza and Steven G. Greenbaum, "Broadband NMR relaxometry as a powerful technique to study molecular dynamics of ionic liquids," ChemPhysChem, 2023
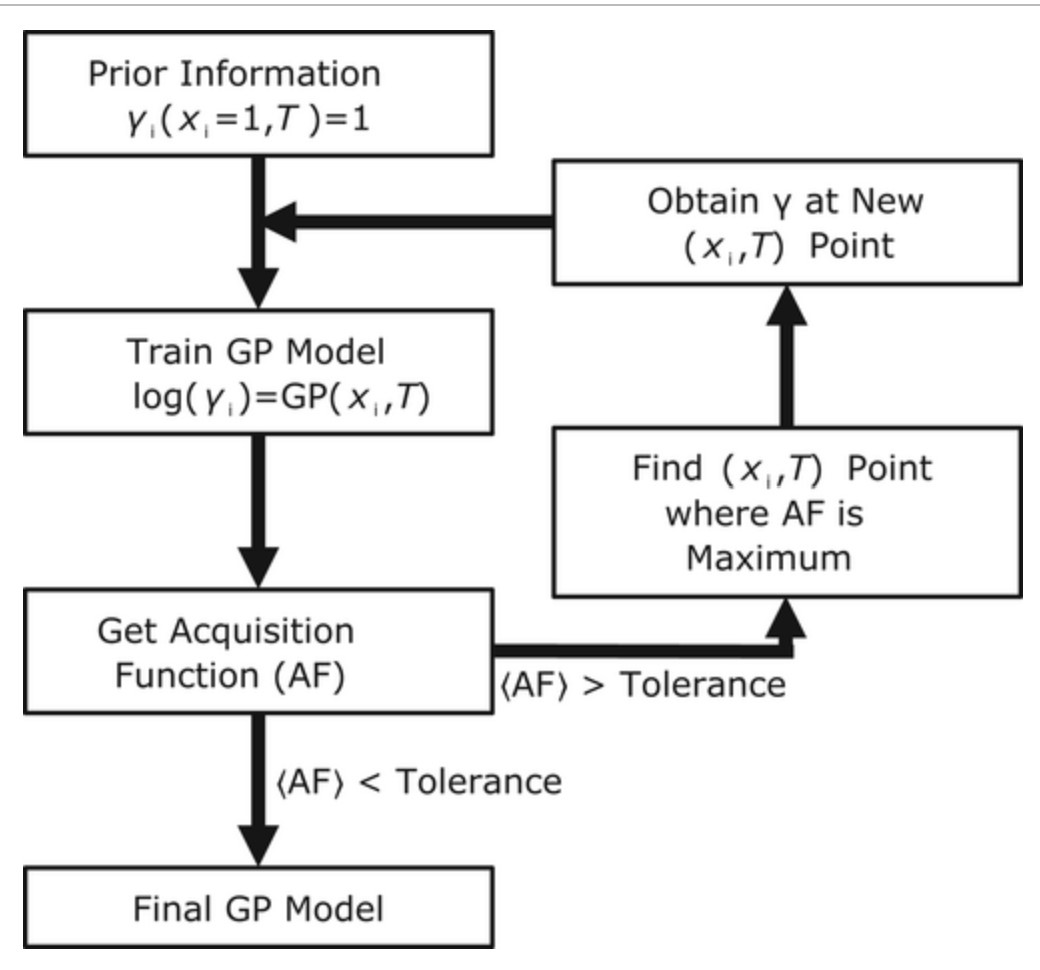
Dinis O. Abraches, Edward J. Maginn, Yamil J. Colón, "Activity Coefficient Acquisition with Thermodynamics-Informed Active Learning for Phase Diagram Construction," AIChE Journal, 2023
DOI: 10.1002/aic.18141
2022
Zhang, Yong; Klein, Jeffrey; Akolkar, Rohan; Gurkan, Burcu; Maginn, Edward, "Solvation Structure, Dynamics and Charge Transfer Kinetics of Cu2+ and Cu+ in Choline Chloride Ethylene Glycol Electrolytes," J. Phys. Chem. B, 2022
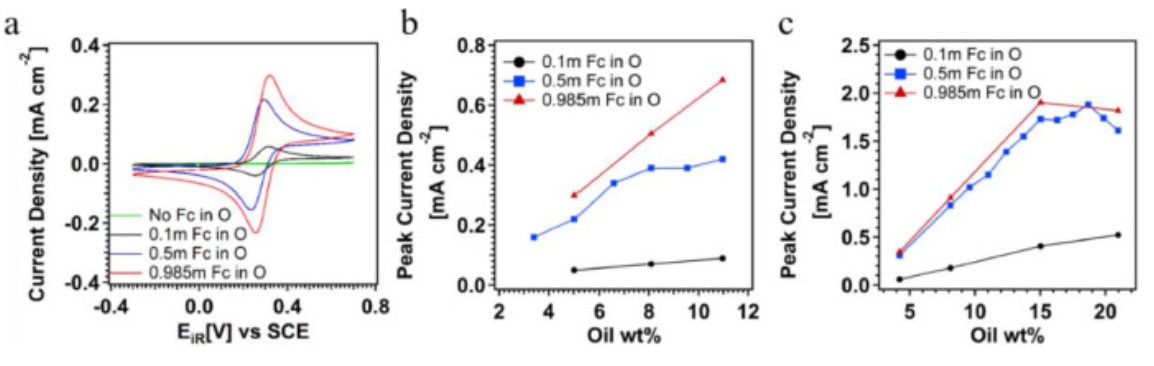
R. Ghahremani, W. Dean, N. Starvaggi, I. Alfurayi, C. Burda, E. Pentzer, J. Wainright, "Redox-Active Eutectic Electrolye with Viologen and Feffocene Derivatives for Flow Battery," ACS Applied Materials and Interfaces, 2022
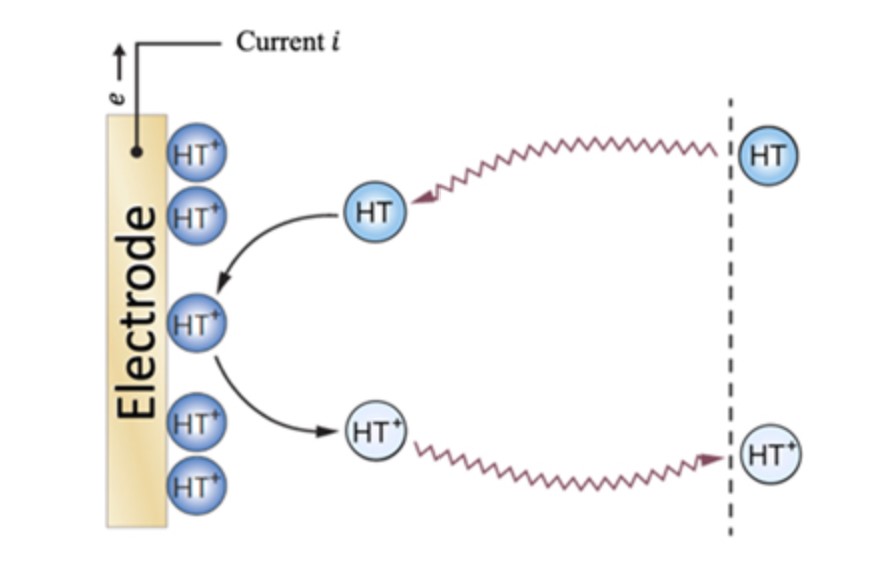
R.Ghahremani, R. Savinell, B. Gurkan, "Perspective-Hydrogen bonded concentrated electrolytes for redox flow batteries: limitations and prospects," J. Electrochemical Society, 2022
A.Imel, B. Barth, D. Hayes,M. Dadmun, T. Zawodzinski, "Microemulsions as Emerging Electrolytes: The Correlation of Structure to Electrochemical Response," ACS Applied Materials and Interfaces, 2022
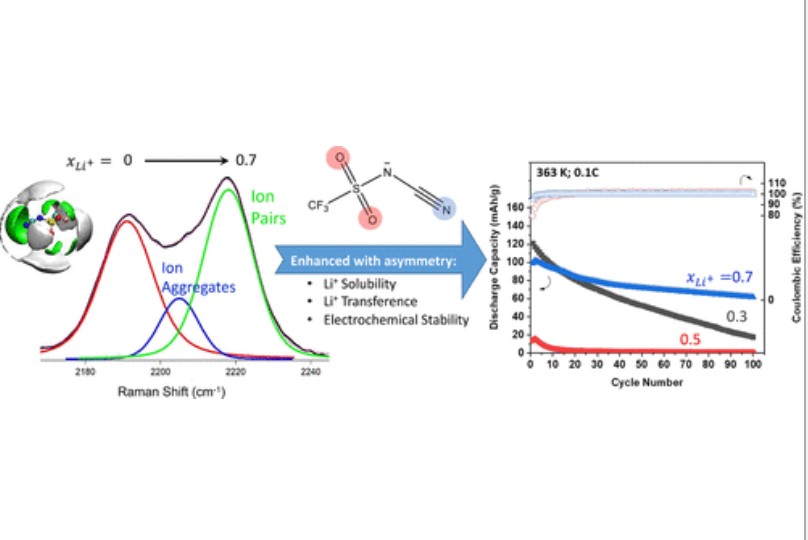
Drace Penley, Xiaoyu Wang, Yun-Yang Lee, Mounesha N Garaga , Raziyeh Ghahremani, Steve Greenbaum, Edward J. Maginn, Burcu Gurkan,"Lithium Solvation and Mobility in Ionic Liquid Electrolytes with Asymmetric Sulfonyl-Cyano Anion," Journal of Chemical and Engineering Data, 2022
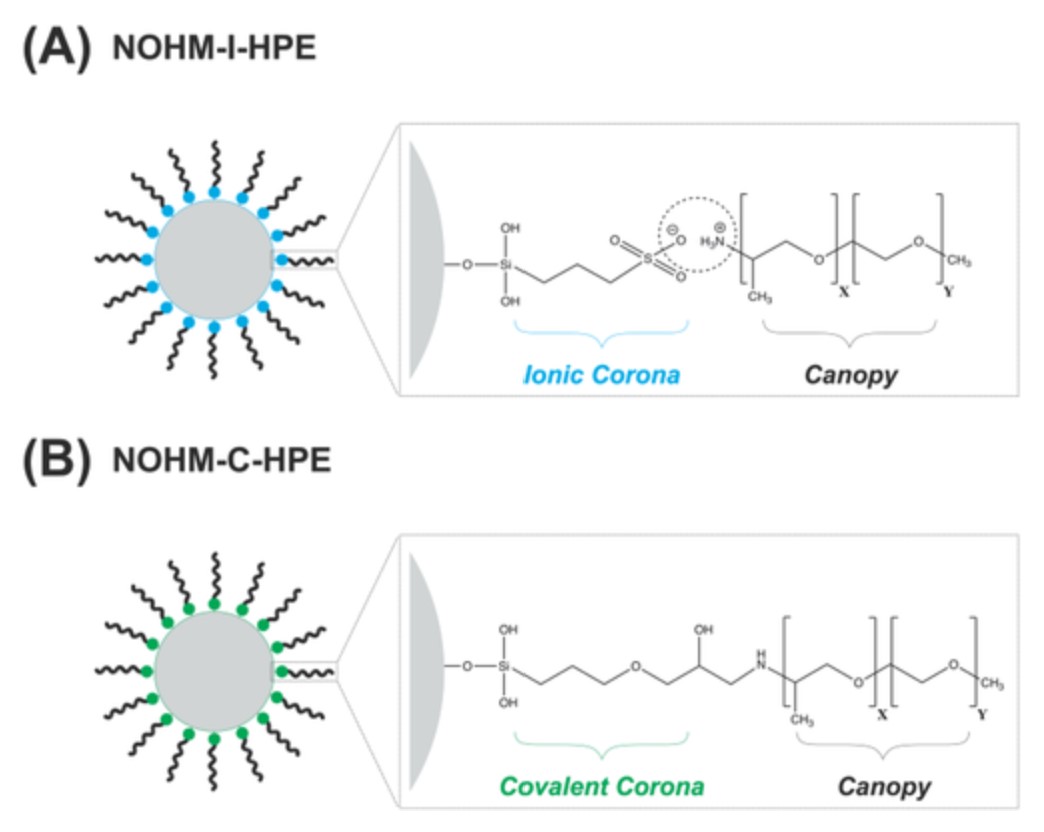
Tony G Feric, Sara T Hamilton, Md Ashraful Haque, Javad Jeddi, Joshua Sangoro, Mark D Dadmun, Ah‐Hyung Alissa Park, "Impacts of Bond Type and Grafting Density on the Thermal, Structural, and Transport Behaviors of Nanoparticle Organic Hybrid Materials-Based Electrolytes," Advanced Functional Materials, 2022
N. Shaheen, W. Dean, D. Penley, B. Kersten, J. Rintamaki, M. Vukmirovic, B. Gurkan, R. Akolkar, "Electro–Oxidation of Nitroxide Radicals: Adsorption–Mediated Charge Transfer Probed Using SERS and Potentiometry," J. Electrochemical Society, 2022
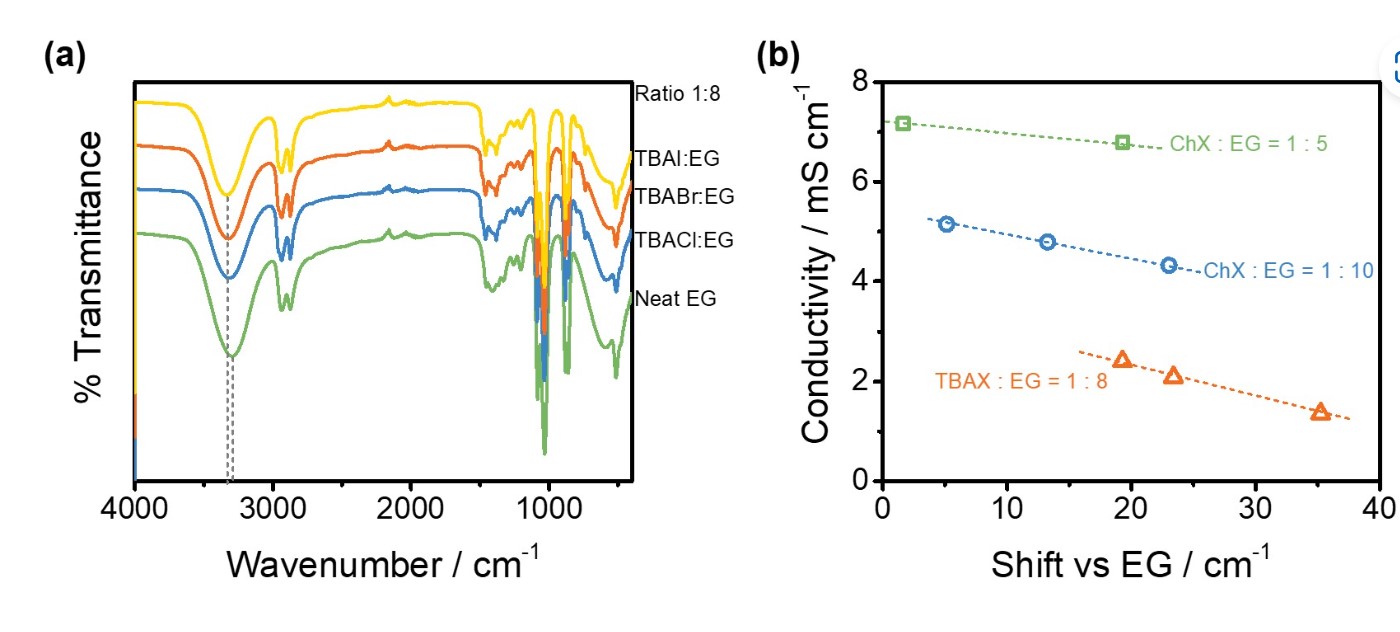
X. Shen, N. Sinclair, C. Kellamis, B. Gurkan, J. Wainright, R. Savinell, "Effects of alkyl chain length and halide anion on hydrogen bonding in eutective solvents," Journal of Molecular Liquids, 2022
DOI: 10.1149/MA2022-02301102mtgabs
Tamar Zelovich, Thorsten Hansen, Mark E. Tuckerman, "A Green's Function Approach for determining Surface Induced Broadening and Shifting of Molecular Energy Levels," Nano Letters, 2022
DOI: 10.1021/acs.nanolett.2c02910
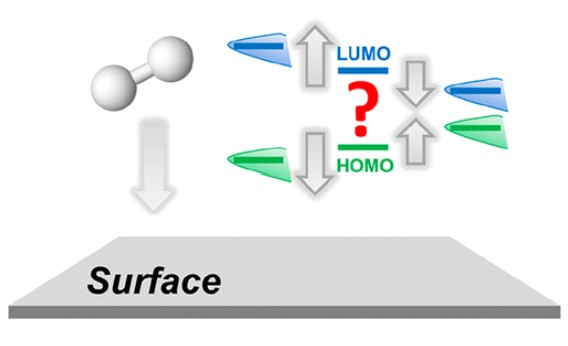
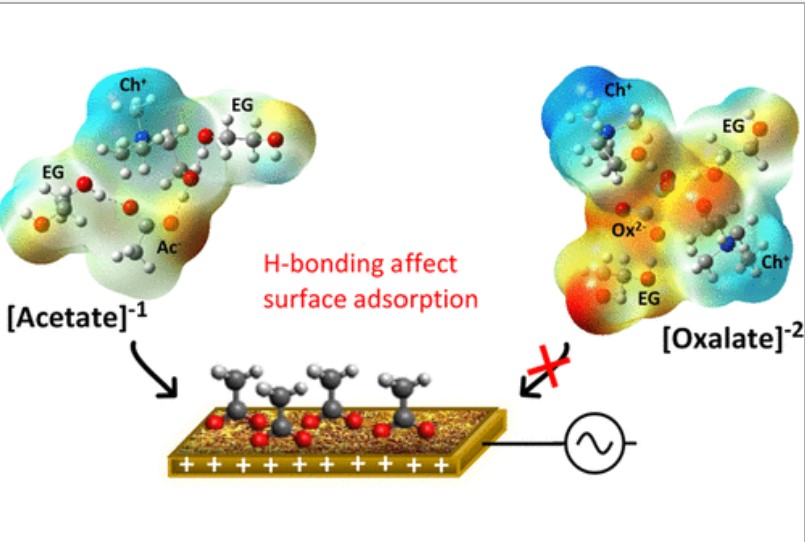
W. Dean, D. Penley, Y. Lee, R. Ghahremani, S. Dongare, B. E. Gurkan, "Anion Effects on the Interfacial Structure and Bulk Physical Properties in Choline-Based Hydrogen-Bonded Electrolytes," The Journal of Physical Chemistry C, 2022.
DOI: 10.1021/acs.jpcc.2c01901
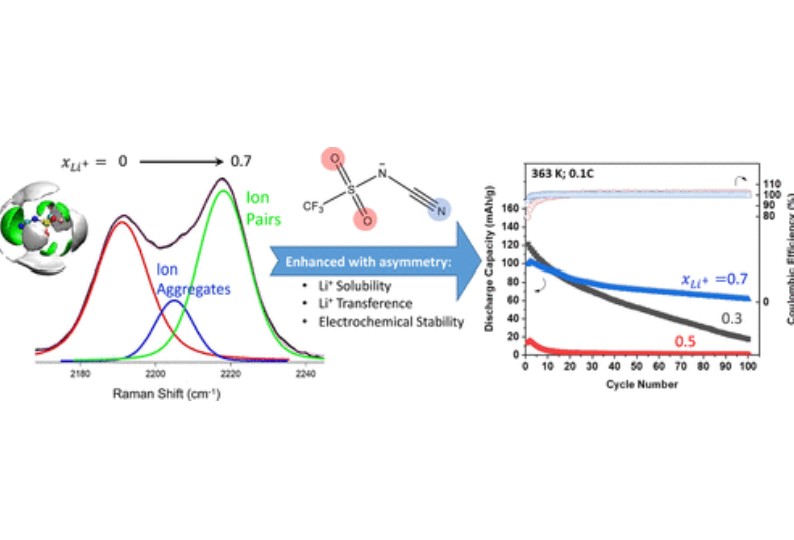
D. Penley, X. Wang, Y. Lee, M. N. Garaga, R. Ghahremani, S. Greenbaum, E. J. Maginn, B. E. Gurkan, "Lithium Solvation and Mobility in Ionic Liquid Electrolytes with Asymmetric Sulfonyl-Cyano Anion," Journal of Chemical & Engineering Data, 2022.
DOI: 10.1021/acs.jced.2c00294
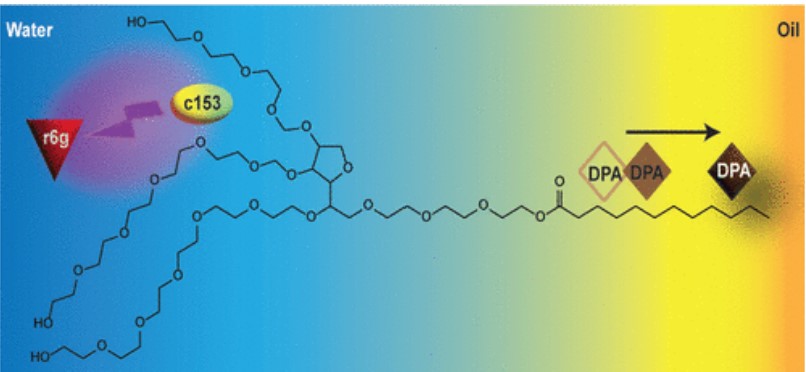
M. R. Hassan, B. A. Colon, J. Russell, T. R. Calhoun, "Small Molecule Sorting: A Fluorescence Study of Microemulsions," J. Phys. Chem. B, 2022.
DOI: 10.1021/acs.
S. T. Hamilton, T. G. Feric, A. Gladysiak, N. M. Cantillo, T. A. Zawodzinski, A. A. Park, "Mechanistic Study of Controlled Zinc Electrodeposition Behaviors Facilitated by Nanoscale Electrolyte Additives at the Electrode Interface," ACS Applied Materials & Interfaces, 2022.
DOI: 10.1021/acsami.1c23781
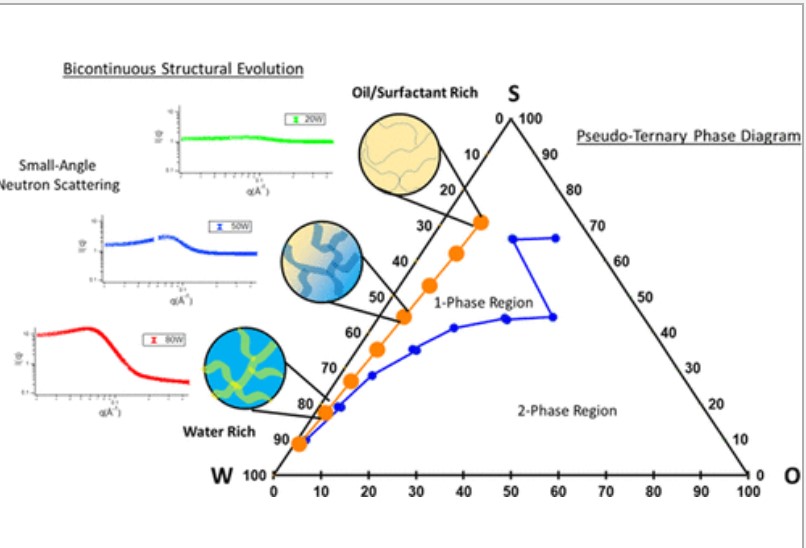
A. E. Imel, B. Barth, D. G. Hayes, M. Dadmun, T. Zawodzinski, "Microemulsions as Emerging Electrolytes: The Correlation of Structure to Electrochemical Response," ACS Applied Materials & Interfaces, 2022.
DOI: 10.1149/1945-
N. Shaheen, W. Dean, J. Rintamaki, M. B. Vukmirovic, B. E. Gurkan, R. Akolkar, "Electro-oxidation of nitroxide radicals: Adsorption-mediated charge transfer probed using SERS and potentiometry," Journal of the Electrochemical Society, 2022.
DOI: 10.1149/1945-
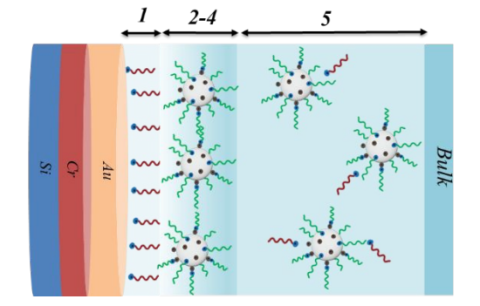
J. M. Klein, H. W. Wang, R. L. Sacci, J. F. Browning, B. Gurkan, "Smooth Modified Surfaces of Silicon for the Study of Ionic Liquid Interfaces by Neutron Reflectometry", ACS Applied Electronic Materials, 2022.
DOI: 10.1021/acsaelm.1c01351
S. T. Hamilton, T. G. Feric, S. Bhattacharyya, N. M. Cantillo, S. G. Greenbaum, T. A. Zawodzinski, A. Park, "Nanoscale Hybrid Electrolytes with Viscosity Controlled Using Ionic Stimulus for Electrochemical Energy Conversion and Storage", JACS Au, 2022.
DOI: 10.1021/jacsau.1c00410
Raziyeh Ghahremani, Robert F. Savinell and Burcu Gurkan, "Hydrogen Bonded Concentrated Electrolytes for Redox Flow Batteries: Limitations and Prospects", Journal of The Electrochemical Society, 2022.
DOI: 10.1149/1945-7111/ac58c6
C. C. Fraenza and S. G. Greenbaum, "Broadband NMR relaxometry of electrolytes for energy storage", Chemical Physics Reviews, 2022.
DOI: 10.1063/5.0076580
Y. Zhang, H. Squire, B. Gurkan, and E. J. Maginn, "Refined Classical Force Field for Choline Chloride and Ethylene Glycol Mixtures over Wide Composition Range", Journal of Chemical & Engineering Data, 2022.
DOI: 10.1021/acs.jced.1c00841
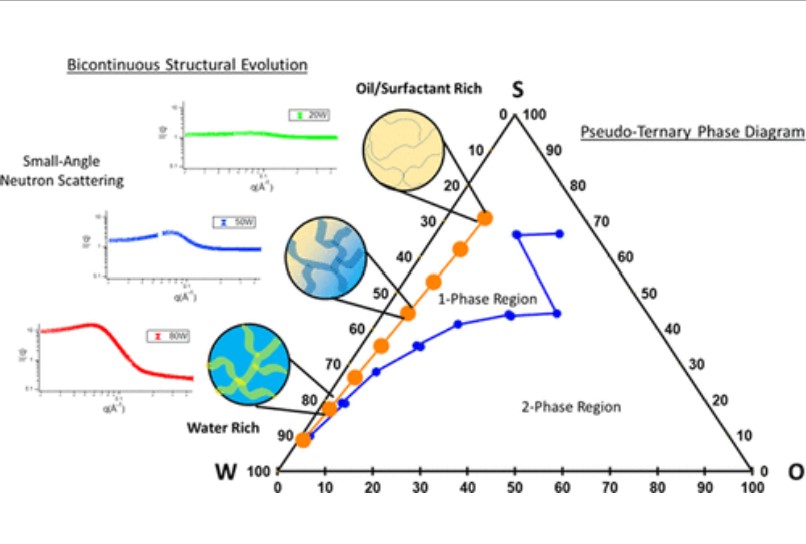
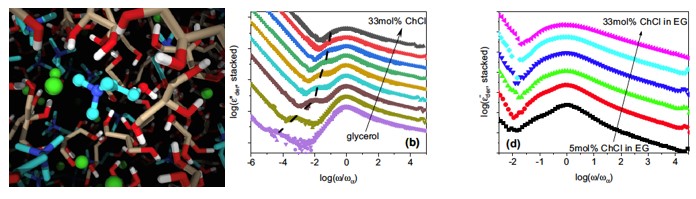
S. Spittle, D. Poe, B. Doherty, C. Kolodziej, L. Heroux, M. A. Haque, H. Squire, T. Cosby, Y. Zhang, C. Fraenza, S. Bhattacharyya, M. Tyagi, J. Peng, R. A. Elgammal, T. Zawodzinski, M. Tuckerman, S. Greenbaum, B. Gurkan, M. Dadmun, E. J. Maginn, and J. Sangoro, "Evolution of Microscopic Heterogeneity and Dynamics in Choline Chloride-based Deep Eutectic Solvents", Nature Communications, 13, 219, 2022.
DOI: 10.1038/s41467-021-27842-z
C. C. Fraenza, R. A. Elgammal, M. N. Garaga, S. Bhattacharyya, T. A. Zawodzinski, and S. G. Greenbaum, "Dynamics of Glyceline and Interactions of Constituents: A Multi-technique NMR study", The Journal of Physical Chemistry B, 126, 4, 890-905, 2022.
DOI: 10.1021/acs.jpcb.1c09227
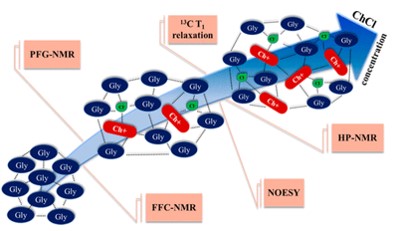
A. E. Imel, B. Barth, D. G. Hayes, M. Dadmun, T. Zawodzinski, "Microemulsions as Emerging Electrolytes: The Correlation of Structure to Electrochemical Response", ACS Applied Materials & Interfaces, 2022.
2021
B. Barth, A. Imel, K. M. Nelms, G. A. Goenaga, T. Zawodzinski, "Microemulsions: Breakthrough Electrolytes for Redox Flow Batteries", Frontiers in Chemistry, 2021.
DOI: 10.1016/j.joule.2020.12.004
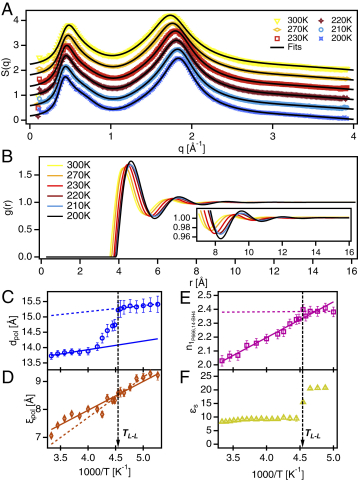
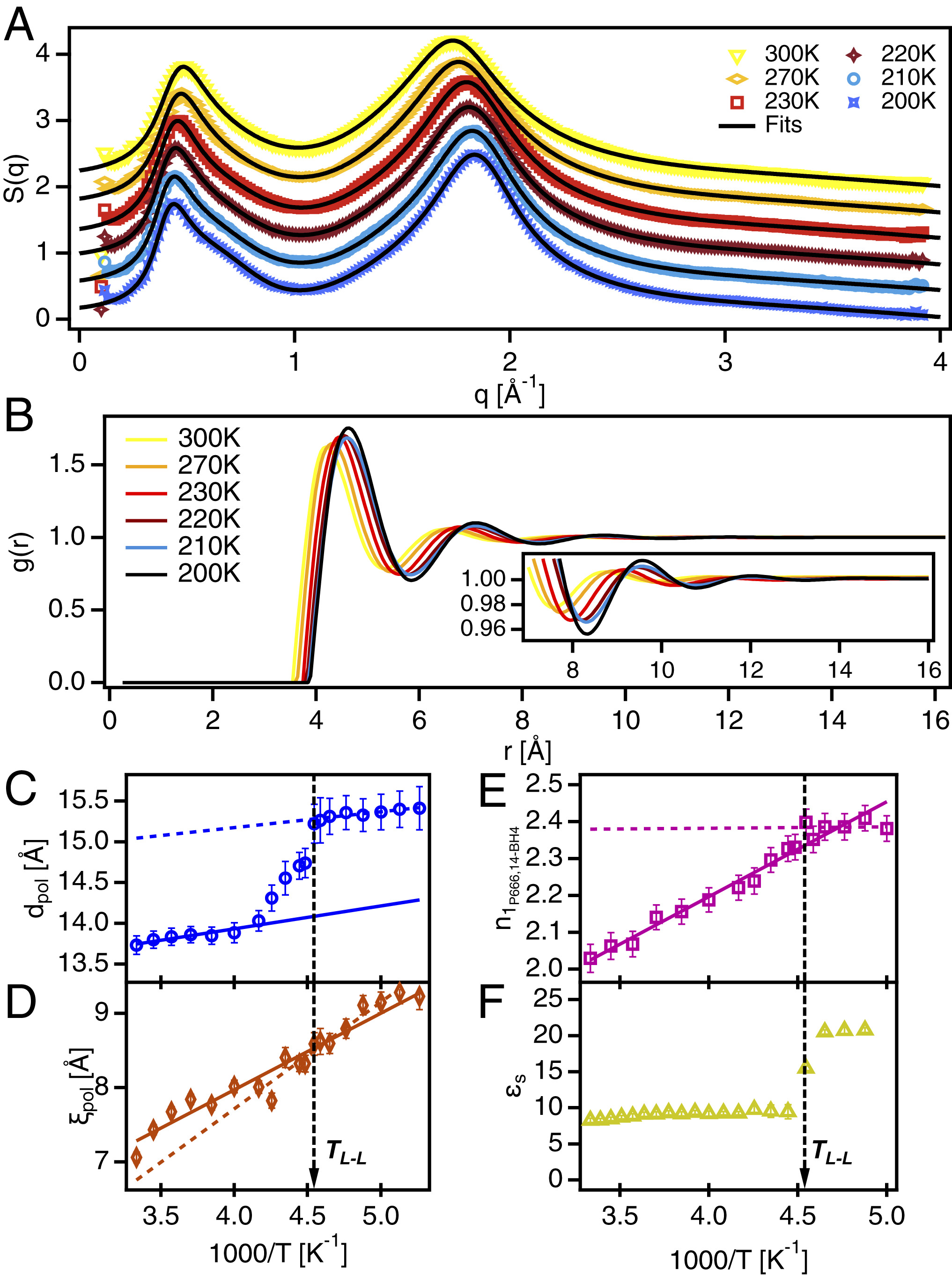
Matthew A. Harris, Thomas Kinsey, Durgesh V. Wagle, Gary A. Baker and Joshua Sangoro, "Evidence of a liquid–liquid transition in a glass-forming ionic liquid", PNAS, 2021.
DOI: 10.1073/pnas.2020878118
J. Peng, Y. Xiao, A. Imel; B. Barth, N. Cantillo, K. M.Nelms, T. A. Zawodzinski, "Electrolyte effects on the electrochemical performance of microemulsions", Electrochimica Acta, 2021.
DOI: 10.1016/j.electacta.2021.139048
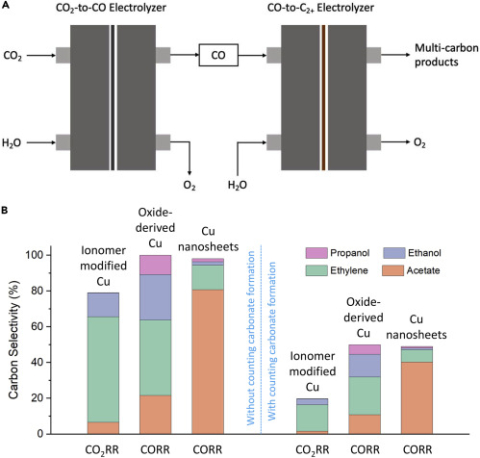
S. Overa, T. G. Feric, A. Park, F. Jiao, "Tandem and Hybrid Processes for Carbon Dioxide Utilization", Joule, 2021.
DOI: 10.1016/j.joule.2020.12.004
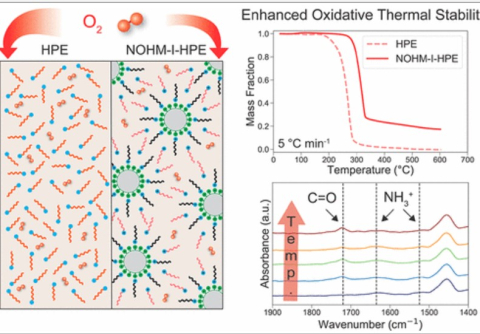
T. G. Feric, S. T. Hamilton, and A. Park, "Insights into the Enhanced Oxidative Thermal Stability of Nanoparticle Organic Hybrid Materials Developed for Carbon Capture and Energy Storage", Energy & Fuels, 2021.
DOI: 10.1021/acs.energyfuels.1c03243
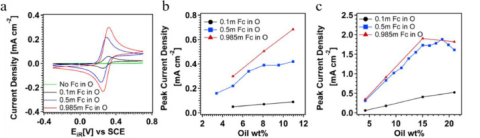
Xiaochen Shen, Nicholas Sinclair, Jesse Wainright, Adam Imel, Brian Barth, Thomas Zawodzinski and Robert F. Savinell, "A Study of Ferrocene Diffusion in Toluene/Tween 20/1-Butanol/Water Microemulsions for Redox Flow Battery Applications", Journal of The Electrochemical Society, 2021.
DOI: 10.1149/1945-7111/ac0b26
X. Shen, N. Sinclair, J. Wainright and R. F. Savinell, "Methods? Analyzing Electrochemical Kinetic Parameters in Deep Eutectic Solvents Using an Extended Butler-Volmer Equation", Journal of The Electrochemical Society, 2021.
DOI: 10.1149/1945-7111/ac006a
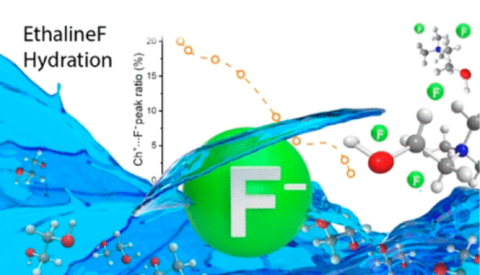
Ibrahim Alfurayj, Carla Cecilia Fraenza, Yong Zhang, Rathiesh Pandian, Stephanie Spittle, Bryce Hansen, William Dean, Burcu Gurkan, Robert Savinell, Steve Greenbaum, Edward Maginn, Joshua Sangoro, and Clemens Burda, "Solvation Dynamics of Wet Ethaline: Water is the Magic Component", The Journal of Physical Chemistry B, 125, 31, 8888-8901, 2021.
DOI: 10.1021/acs.jpcb.1c04629

Jing Peng, Nelly M. Cantillo, Ye Xiao, K. McKensie Nelms, Lacey S. Roberts, Gabriel Goenaga, Adam Imel, Brian Andrew Barth, Mark Dadmun, Douglas G. Hayes and Thomas Zawodzinski, "Decoupling Conductivity and Solubility in Electrolytes Using Microemulsions", Journal of The Electrochemical Society, 2021.
DOI: 10.1149/1945-7111/ac180b
Nicholas S. Sinclair, Xiaochen Shen, Evan Guarr, Robert F. Savinell and Jesse S. Wainright, "Electrochemical Decomposition of Primary Alcohol Groups in Deep Eutectic Solvents", Journal of The Electrochemical Society, 2021.
DOI: 10.1149/1945-7111/ac2d14
Tony G. Feric, Sara T. Hamilton, Nelly M. Cantillo, Adam E. Imel, Thomas A. Zawodzinski, and Ah-Hyung Alissa Park, "Dynamic Mixing Behaviors of Ionically Tethered Polymer Canopy of Nanoscale Hybrid Materials in Fluids of Varying Physical and Chemical Properties", The Journal of Physical Chemistry B, 2021.
DOI: 10.1021/acs.jpcb.1c00935
Mounesha N. Garaga, Nishani Jayakody, Carla C. Fraenza, Boris Itin, Steven Greenbaum, "Molecular-level insights into structure and dynamics in ionic liquids and polymer gel electrolytes", Journal of Molecular Liquids, 2021.
DOI: 10.1016/j.molliq.2021.115454
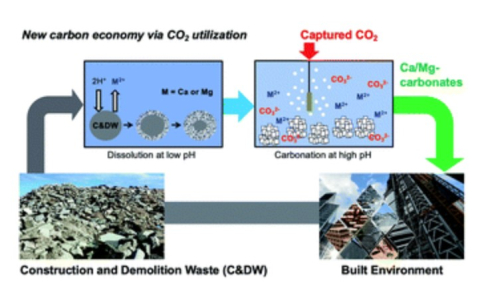
Guanhe Rim, Noyonika Roy, Diandian Zhao, Shiho Kawashima, Phillip E. Stallworth, Steve G. Greenbaum and Ah-Hyung Alissa Park , "CO2 Utilization in Built Environment via the PCO2 Swing Carbonation of Alkaline Solid Wastes with Different Mineralogy", Faraday Discussions, 2021.
DOI: 10.1039/D1FD00022E
Md Ashraful Haque, Tony G. Feric, Sara T. Hamilton, Ah-Hyung Alissa Park, Mark D. Dadmun, "Structure and Dispersion of Free and Grafted Polymer in Nanoparticle Organic Hybrid Materials-based Solutions by Small Angle Neutron Scattering", The Journal of Physical Chemistry C, 2021.
DOI: 10.1021/acs.jpcc.0c10790
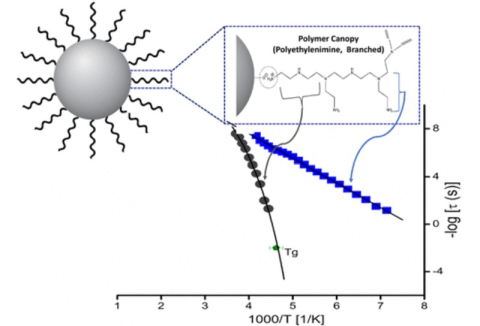
Emmanuel Urandu Mapesa, Nelly M. Cantillo, Sara Triana Hamilton, Matthew A. Harris, Thomas A. Zawodzinski Jr., Ah-Hyung Alissa Park, Joshua Sangoro, "Localized and Collective Dynamics in liquid-like Polyethylenimine-based Nanoparticle Organic-Inorganic Hybrid Materials", Macromolecules, 2021.
DOI: 10.1021/acs.macromol.0c02370
Nicholas S. Sinclair, Derrick Poe, Robert F. Savinell, Edward J. Maginn, Jesse S. Wainright, "A Nitroxide Containing Organic Molecule in a Deep Eutectic Solvent for Flow Battery Applications ", J. Electrochem. Soc., 168, 020527, 2021.
DOI: 10.1149/1945-7111/abe28a
William Dean, Jeffrey Klein and Burcu Gurkan, "Do Deep Eutectic Solvents Behave Like Ionic Liquid Electrolytes? A Perspective from the Electrode-Electrolyte Interface2" Journal of The Electrochemical Society, 168, 026503, 2021.
DOI: 10.1149/1945-7111/abde83
2020
B. E. Gurkan, E. J. Maginn, and E. B. Pentzer, "Deep Eutectic Solvents: A New Class of Versatile Liquids", The Journal of Physical Chemistry B, 2020.
DOI: 10.1021/acs.jpcb.0c10099
Yun-Yang Lee, Drace Penley, Aidan Klemm, William Dean, and Burcu Gurkan, "Deep Eutectic Solvent Formed by Imidazolium Cyanopyrrolide and Ethylene Glycol for Reactive CO 2 Separations" ACS Sustainable Chemistry Engineering, 2020.
DOI: 10.1021/acssuschemeng.0c07217
Hansen, B. B.; Spittle, S.; Chen, B.; Poe, D.; Zhang, Y.; Klein, J. M.; Horton, A.; Adhikari, L.; Zelovich, T.; Doherty, B. W.; Gurkan, B.; Maginn, E. J.; Ragauskas, A.; Dadmun, M.; Zawodzinski, T. A.; Baker, G. A.; Tuckerman, M. E.; Savinell, R. F.; Sangoro, J. R., "Deep Eutectic Solvents: A Review of Fundamentals and Applications", Chemical Review, 121, 3, 1232-1285, 2020.
DOI: 10.1021/acs.chemrev.0c00385
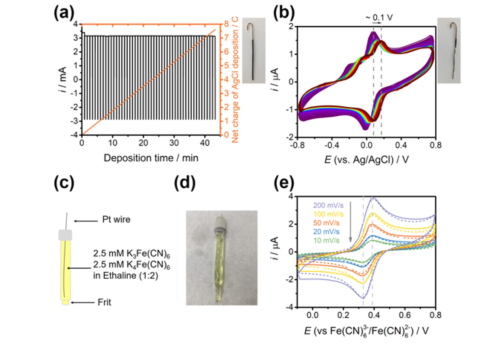
Nora A. Shaheen,Mahesh Ijjada, Miomir B. Vukmirovic, and Rohan Akolkar, "Mechanism of Electrochemical Oxidation of Nitroxide Radicals in Ethaline Deep Eutectic Solvent" J. Electrochem. Soc., 167, 143505, 2020.
DOI: 10.1149/1945-7111/abc439
Jing Peng, Nelly M. Cantillo, K. McKensie Nelms, Lacey S. Roberts, Gabriel Goenaga, Adam Imel, Brian Andrew Barth, Mark Dadmun, Luke Heroux, Douglas G. Hayes, and Thomas Zawodzinski, "Electron Transfer in Microemulsion-Based Electrolytes" ACS Applied Materials and Interfaces, 2020.
DOI: 10.1021/acsami.0c07028
Nelly M. Cantillo, Maria Bruce, Sara T. Hamilton, Tony G. Feric, Ah-Hyung Alissa Park and Thomas A. Zawodzinski Jr., "Electrochemical Behavior of Copper Ion Complexed with Nanoparticle Organic Hybrid Materials" IOP Science, 2020.
DOI: 10.1149/1945-7111/aba158
Nishani K Jayakody, Carla C. Fraenza, Steven G. Greenbaum, David S. Ashby, and Bruce S. Dunn, "NMR Relaxometry and Diffusometry Analysis of Dynamics in Ionic Liquids and Ionogels for Use in Lithium Ion Batteries" Journal of Physical Chemistry B, 2020.
DOI: 10.1021/acs.jpcb.0c02755
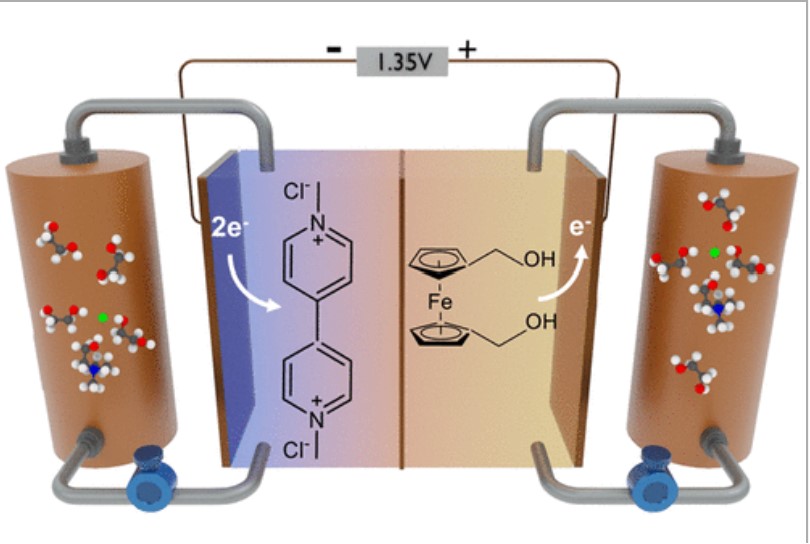
Brian Chen, Sarah Mitchell, Nicholas Sinclair, Jesse Wainright, Emily Pentzer and Burcu Gurkan, "Feasibility of TEMPO-functionalized imidazolium, ammonium and pyridinium salts as redox-active carriers in ethaline deep eutectic solvent for energy storage", Molecular Systems Design and Engineering, 2020.
DOI: 10.1039/D0ME00038H
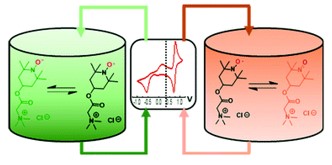
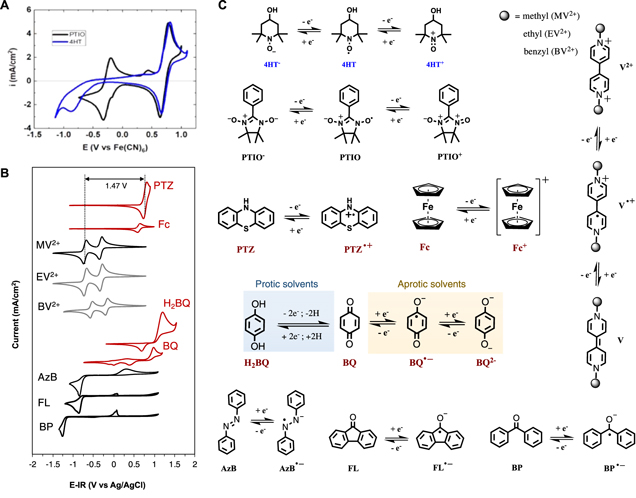
Jeffrey M. Klein, Henry Squire, William Dean, and Burcu E. Gurkan, "From Salt in Solution to Solely Ions – Solvation of MethylViologen in Deep Eutectic Solvents and Ionic Liquids", Journal of Physical Chemistry B, 2020.
DOI: 10.1021/acs.jpcb.0c03296
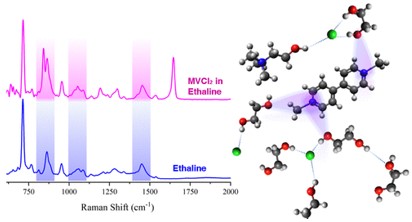
Yong Zhang, Derrick Poe, Luke Heroux, Henry Squire, Brian W. Doherty, Zhuoran Long,Mark Dadmun, Burcu Gurkan, Mark E. Tuckerman, and Edward J. Maginn, "Liquid Structure and Transport Properties of the Deep EutecticSolvent Ethaline", Journal of Physical Chemistry B, 124, 5251-5264, 2020.
DOI: 10.1021/acs.jpcb.0c04058
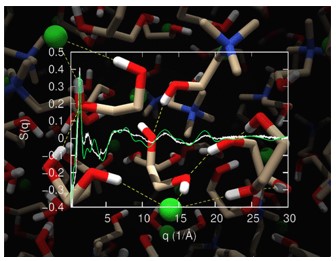
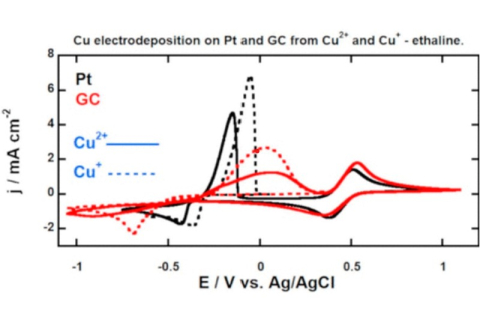
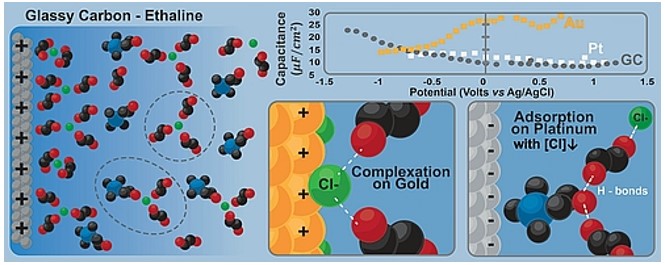
Miomir B. Vukmirovic, Radoslav R. Adzic, and Rohan Akolkar, "Copper Electrodeposition from Deep Eutectic Solvents – Voltammetric Studies Providing Insights into the Role of Substrate: Platinum vs. Glassy Carbon", Journal of Physical Chemistry B, 2020.
DOI: 10.1021/acs.jpcb.0c02735
X. Shen, N. Sinclair, J. Wainright, R. Akolkar, and R.F. Savinell, "Evaluating and developing a reliable reference electrode for choline chloride based deep eutectic solvents", Journal of the Electrochemical Society, 2020 167 (8).
DOI: 10.1149/1945-7111/ab913c
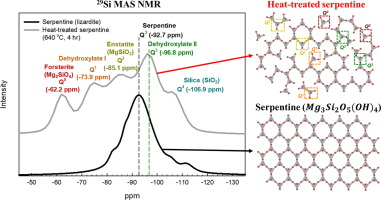
Guanhe Rim, Ariane Katrina Marchese, Phillip Stallworth, Steven G. Greenbaum, Ah-Hyung Alissa Park, "29 Si Solid State MAS NMR Study on Leaching Behaviors and Chemical Stability of Different Mg-Silicate Structures CO2 Sequestration", Chemical Engineering Journal, 396 (c).
DOI: 10.1016/j.cej.2020.125204
L. Chang and A. J. Bard, "Electrochemical Characterization of Bromine Reduction to Tribromide in Individual Nitrobenzene-in-Water Emulsion Droplets", Journal of The Electrochemical Society, 2020 167 (6).
DOI: 10.1149/1945-7111/ab80ab
Nora A. Shaheen, Ijjada Mahesh, Miomir B. Vukmirovic, Rohan Akolkar, "Hysteresis effects and roughness suppression efficacy of polyethylenimine additive in Cu electrodeposition in ethaline", Electrochemistry Communications 2020 115 106721
DOI: 10.1016/j.elecom.2020.106721
2019
J.M. Klein; H. Squire; B. Gurkan, "Electroanalytical Investigation of the Electrode–Electrolyte Interface of Quaternary Ammonium Ionic Liquids: Impact of Alkyl Chain Length and Ether Functionality, The Journal of Physical Chemistry C, 124, 10, 5613-5623, (2019).
DOI: 10.1021/acs.jpcc.9b08016
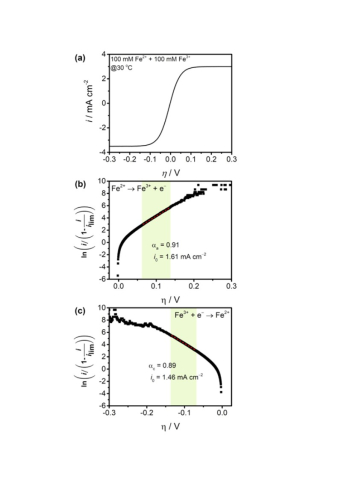
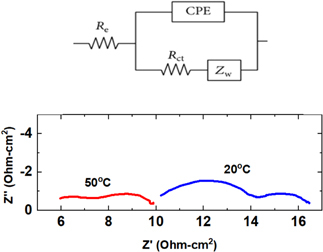
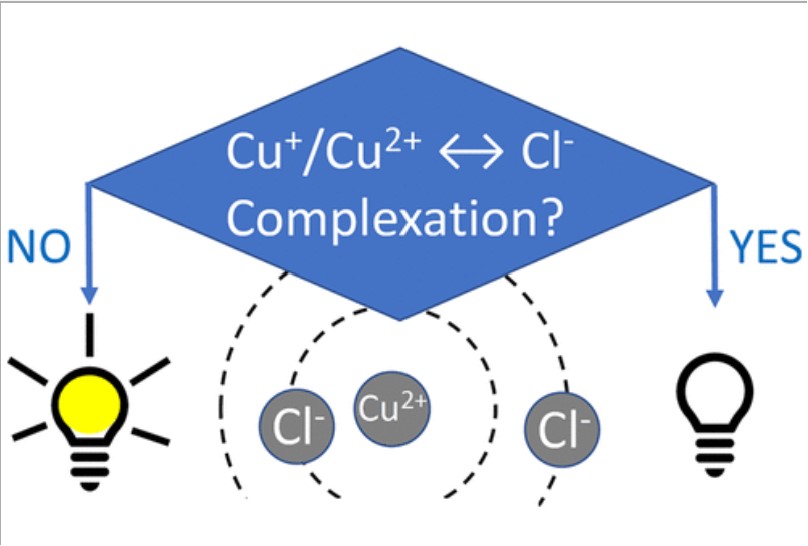
Dai Shen, Miomir B Vukmirovic, Rohan Akolkar, "Understanding the Role of Complexation in the Charge-Transfer Kinetics of the Cu2+ + e ↔ Cu1+ Redox Reaction in Ethaline Deep Eutectic Solvent", Journal of The Electrochemical Society, 166 (15), E526-E532 (2019)
DOI: 10.1149/2.1211915jes
Burcu Gurkan, Henry Squire, and Emily Pentzer, "Metal-Free Deep Eutectic Solvents: Preparation, Physical Properties, and Significance", The Journal of Physical Chemistry Letters 2019 10 (24), 7956-7964 (2019)
DOI: 10.1021/acs.jpclett.9b01980

2018
Dai Shen, Katherine Steinberg, Rohan Akolkar, “Avoiding Pitfalls in the Determination of Reliable Electrochemical Kinetics Parameters for the Cu2+→Cu1+ Reduction Reaction in Deep Eutectic Solvents”, Journal of The Electrochemical Society , 165 (14), E808-E815, 2018,
DOI: 10.1149/2.1011814jes