Our mission is to uncover the transport mechanisms of ions, protons, redox species, and electrons in nano to meso scale structured electrolytes in the bulk and at the electrode-electrolyte interfaces to achieve high energy and power density in next generation energy storage systems.
BEES2 Research Impacts Many Fields.
Motivation
Next generation energy storage and chemical transformation technologies require major advances in electrolytes discovery to achieve safer and more efficient performance. Such advances, or breakthroughs, can be in properties or mechanisms not realized in conventional electrolytes. The BEES (Breakthrough Electrolytes for Energy Storage) EFRC, in its first four years (2018 to 2022), studied novel benign electrolytes that are structured at the molecular to meso-scale level. A fundamental understanding of the physical, transport, and electrochemical properties of representative systems in relation to their bulk and interfacial structures was achieved through collaborative experimental and theoretical studies within BEES.
In the EFRC BEES2 (Breakthrough Electrolytes for Energy Storage Systems) the overarching goal is to further define design principles of structured electrolytes for achieving breakthroughs in energy density and transport rates of redox species and ions for large scale energy storage devices such as redox flow batteries which are critical to store energy produced from carbon neutral sources such as sunlight, wind, and other renewables.
BEES2 Research Enables Next Generation Energy Storage Systems.
Strategy
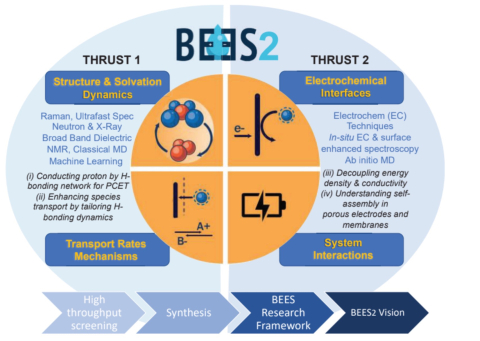
BEES2 employs a strategy that leverages electrolyte structure to (i) conduct protons for proton coupled electron transfer reactions; (ii) enhance species transport in the bulk and at interfaces; (iii) decouple energy density from conductivity; and (iv) control self-assembly in porous electrodes and membranes by expanded efforts in synthesis, computations, and high through-put screening.
Thrust 1: Solvation, Dynamics, and Transport
In Thrust 1, the main aim is to understand solvation and the coupling of the structural effects of moving ions/protons in the bulk with the dynamics of the transport. Developing these relations requires a hierarchy of experimental and computational approaches that have been established by the BEES team and this established research framework will accelerate discovery of breakthrough electrolytes in BEES2 that are concentrated yet sufficiently conductive.
Thrust 2: Interfacial Structure and Electron Transfer
In Thrust 2, the main aim is to understand the voltage induced processes at the electrode-electrolyte interface that include electrolyte interfacial structuring, electrochemical stability, electron transfer mechanism, and kinetics. The understanding of the interfacial redox reactions cannot be complete without an established link to the solvation structure, dynamics, and transport.