In general, my laboratory looks at understanding how biomaterials and cell-based therapeutics can be designed to correct suppressive immune responses in solid tumor cancers. My aim is to develop immunoengineering technology (biomaterials, drug delivery system, and nanotechnology) to shift the balance of the immune response between activation and suppression. Where possible, my hope is to exploit research discovery to improve patient outcomes by enhancing responses to conventional, targeted or immunotherapeutic. Currently, the laboratory focuses primarily on pancreas cancer, advanced melanoma and osteosarcoma.
Y-Shaped Peptide Targeting Tumor-Associated Macrophages (TAMs)
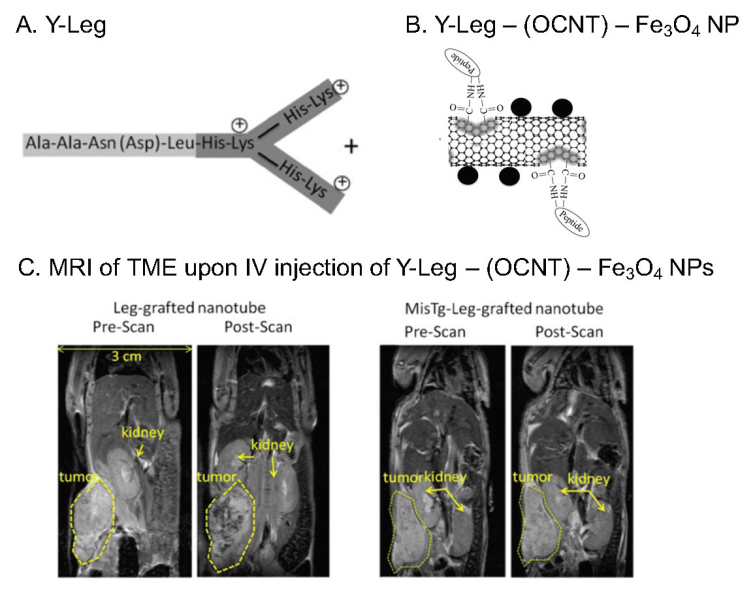
TAM is increasingly being viewed as a target of great interest in TME due to its important role in the progression and metastasis of cancers. It has been shown that TAM indeed overexpresses unique surface marker legumain. In our study, we designed and synthesized a Y-shaped legumain-targeting peptide (Y-Leg) with functional groups allowing for further conjugation with imaging and therapeutic moieties (vide infra) (Figure 1 A). The in vitro cell experiments using FITC-conjugated Y-Leg revealed its specific and selective interaction with M2-polarized macrophages (i.e., TAM) with preference to M1 macrophages, and that the interaction was not interfered with by conjugating FITC to its functional groups.;
Drug Deliver System for Therapeutic Imaging
Further, we constructed a nanotube system by grafting Y-Leg onto oxidized carbon nanotubes (OCNTs) loaded with paramagnetic Fe3O4 nanoparticles (Figure 1 B). The intravenous injection to 4T1 mammary tumor-bearing mouse led to magnetic resonance imaging of TME (Figure 1 C). Importantly, the Y shape of peptide and its functional groups containing amines and imidazole can protonate at different pHs, contributing to the in vitro and in vivo targeting specificity. The methodology developed in our study is applicable to the construction of various multifunctional nanoparticle systems for selectively targeting, imaging and manipulating of TAMs for the diagnosis and treatment of cancers and inflammatory diseases identified with macrophage infiltrated disease tissue.
Biomolecule BG34-200 Inducing myeloid cell activation and re-programming TAMs in TME
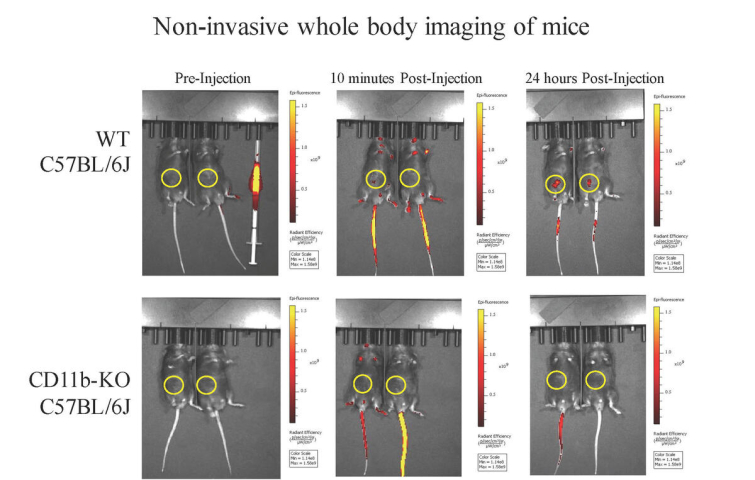
Figure 2. IV Administration of AF647-tagged BG34-200
Induces Tumor Accumulation dependent on CD11b
Zhang et al., IJBCB, 2012, V44, 914-927
Zhang et al, OncoImmunology, 2018, 7: 2.
Zhang et al, Frontiers in Immunology, 2018, 9, 341
Cancer immunotherapies promote immune-mediated control of tumor growth and metastasis by mounting tumor-reactive T cell responses. However, clinical success has been limited as the tumor microenvironment is infiltrated with immunosuppressive myeloid cells and tumor promoting M2 macrophages that drive T cell exclusion and dysfunction.
Our team has recently developed BG34-200, an oat bran derived beta-(1,3)-(1,4)-glucans (BG34) with well-characterized molecular weight (Mw, 200 kda) and chemical structure. We have prepared BG34 of different Mw and performed non-clinical studies that have demonstrated: 1) the in vivo antitumor responses can be effectively induced when highly purified BG34 of 200 kDa (BG34-200) was used; 2) BG34-200 mediates robust antitumor responses in mice bearing highly metastatic osteosarcoma (OS) with non-immunogenic tumor microenvironment (TME), resulting in striking regression of tumor growth and metastasis; 3) IV administration appears to be the optimal method of treatment; 4) the BG34-200 molecule processes an unusual capacity to enter monocyte and macrophage lineage via CD11b (CR3: CD11b/CD18; Mac-1) (Figure 2) and trigger functional polarization towards immune-stimulating M1 differentiation; 5) BG34-200 treatment associates with rapid myeloid cell mobilization and M1 macrophage activation both in vitro and in vivo, suggesting its ability to directly or indirectly induce “trained immunity” with the acquisition of memory responses. In summary, our data support a model in which BG34-200 interacts directly with CD11b to impact its modulation on myeloid cell states with improved antigen presenting capacity in vitro and increased dendritic cells (DCs) and T cell subsets prevalence within TME, accompanied by a reduction of tumor growth/metastasis without overt toxicity or progressive efficacy loss. We have filed patents for BG34-200 and are working with Case TTO to push this molecule to clinical application.